Acids and Basis
Acids and bases are two fundamental categories of substances in chemistry, with distinct properties and behaviors. Here's a brief overview of each:
Acids:
- Acids are substances that donate protons (H⁺ ions) when dissolved in water.
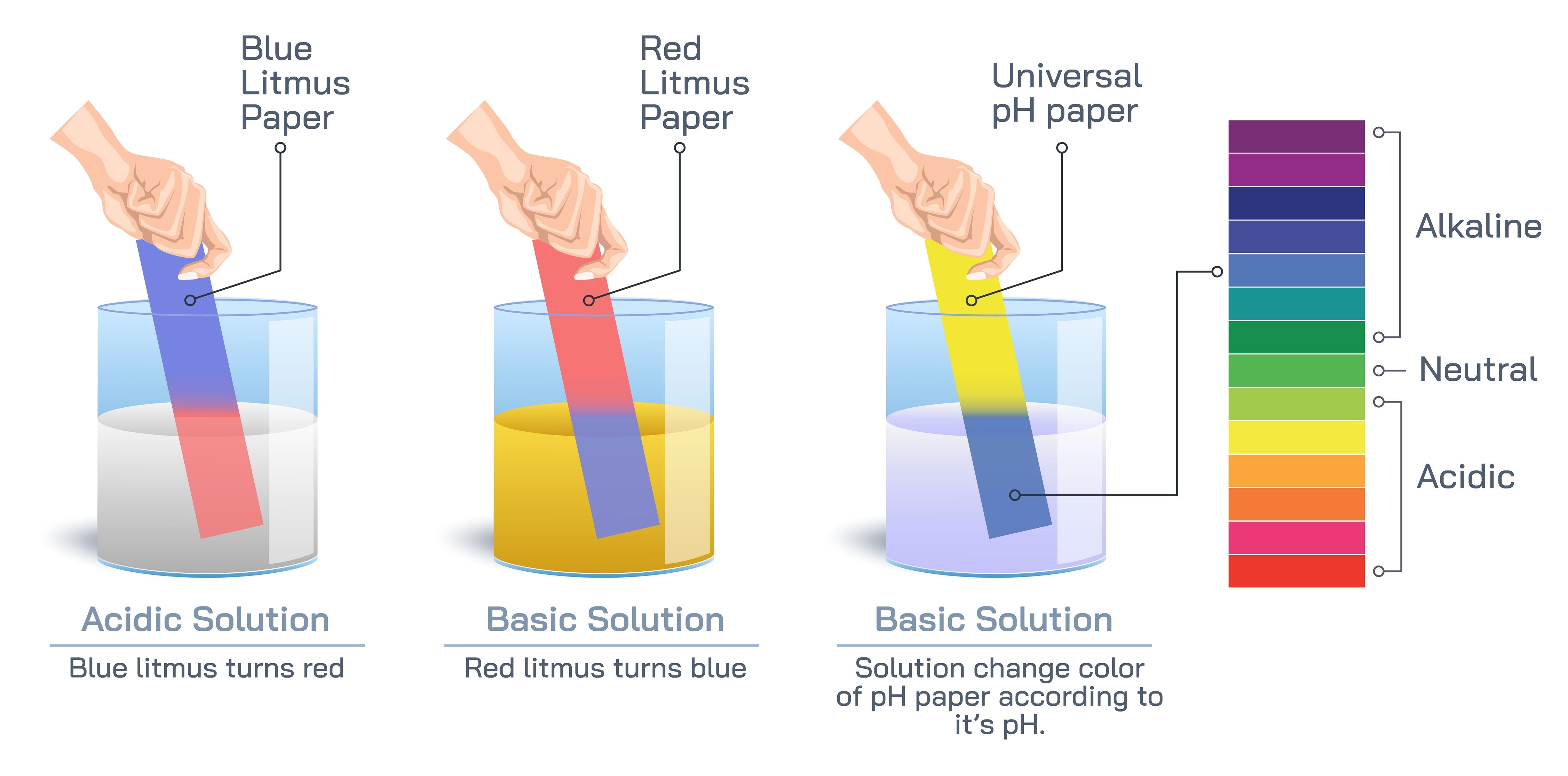
- They have a sour taste and can turn blue litmus paper red.
- Examples of common acids include hydrochloric acid (HCl), sulfuric acid (H₂SO₄), citric acid (found in citrus fruits), and acetic acid (found in vinegar).
- Acids react with metals to produce hydrogen gas and with carbonates to produce carbon dioxide gas.
- Acids have a pH less than 7. The lower the pH, the stronger the acid.
Bases:
- Bases are substances that accept protons (H⁺ ions) or donate hydroxide ions (OH⁻) when dissolved in water.
- They typically have a bitter taste and feel slippery to the touch.
- Bases turn red litmus paper blue.
- Examples of common bases include sodium hydroxide (NaOH), potassium hydroxide (KOH), and ammonia (NH₃).
- Bases react with acids to form water and a salt in a reaction called neutralization.
- Bases have a pH greater than 7. The higher the pH, the stronger the base.
pH Scale:
- The pH scale is a measure of the acidity or basicity of a solution. It ranges from 0 to 14.
- A pH of 7 is considered neutral, meaning the concentration of H⁺ ions equals the concentration of OH⁻ ions.
- pH values less than 7 indicate acidic solutions, with lower values indicating stronger acidity.
- pH values greater than 7 indicate basic (or alkaline) solutions, with higher values indicating stronger basicity.
Understanding acids and bases is crucial in various fields, including chemistry, biology, environmental science, and medicine, as they play significant roles in chemical reactions, industrial processes, and biological systems.

Here are some common examples of acids:
- Hydrochloric Acid (HCl): Hydrochloric acid is a strong acid found naturally in the stomach, where it aids in the digestion of food. It is also widely used in industry for various purposes, including cleaning and pickling.
- Sulfuric Acid (H2SO4): Sulfuric acid is one of the most widely used industrial chemicals, with applications ranging from battery production to fertilizer manufacturing. It is a strong acid and highly corrosive.
- Citric Acid: Citric acid is a weak organic acid found naturally in citrus fruits such as lemons, oranges, and limes. It is commonly used as a food additive to impart a sour taste or as a preservative.
- Acetic Acid: Acetic acid, also known as vinegar when diluted in water, is a weak acid with a distinctive sour taste and pungent odor. It is used in food preparation, pickling, and as a household cleaning agent.
- Phosphoric Acid (H3PO4): Phosphoric acid is commonly used in the food and beverage industry as an acidulant in soft drinks, providing a tangy flavor. It is also used in rust removal and as a chemical reagent.
- Nitric Acid (HNO3): Nitric acid is a strong acid used primarily in the production of fertilizers, explosives, and various organic compounds. It is highly corrosive and can cause severe burns upon contact with skin.
These are just a few examples of acids, but there are many others with diverse properties and applications in chemistry, industry, and everyday life. Here are some common examples of bases:
- Sodium Hydroxide (NaOH): Sodium hydroxide, also known as caustic soda or lye, is a strong base commonly used in industry for manufacturing soaps, detergents, and various chemicals. It is highly corrosive and can cause severe burns upon contact with skin.
- Potassium Hydroxide (KOH): Potassium hydroxide is another strong base similar to sodium hydroxide. It is used in the production of fertilizers, detergents, and pharmaceuticals, as well as in the manufacture of batteries and other industrial processes.
- Ammonia (NH3): Ammonia is a weak base commonly used in household cleaning products, as well as in fertilizers and various industrial processes. It has a pungent odor and is highly soluble in water.
- Calcium Hydroxide (Ca(OH)2): Calcium hydroxide, also known as slaked lime, is a white powder produced by mixing calcium oxide (quicklime) with water. It is used in construction, agriculture, and wastewater treatment, as well as in the production of certain foods like tortillas and pickles.
- Magnesium Hydroxide (Mg(OH)2): Magnesium hydroxide is a white powder commonly used as an antacid to neutralize stomach acid and relieve heartburn, indigestion, and constipation. It is also used in the production of magnesium salts and as a fire retardant.
- Aluminum Hydroxide (Al(OH)3): Aluminum hydroxide is a white gel-like substance used as an antacid to treat symptoms of acid indigestion and heartburn. It is also used as a flame retardant in plastics and as a filler in cosmetics and pharmaceuticals.
These are just a few examples of bases, but there are many others with diverse properties and applications in chemistry, industry, and everyday life.