IONS In Chemistry
In chemistry, ions are electrically charged particles formed when atoms gain or lose electrons. Atoms typically have an equal number of protons (positively charged particles) and electrons (negatively charged particles), resulting in a neutral charge overall. However, when an atom gains or loses one or more electrons, it becomes an ion with a net electrical charge.
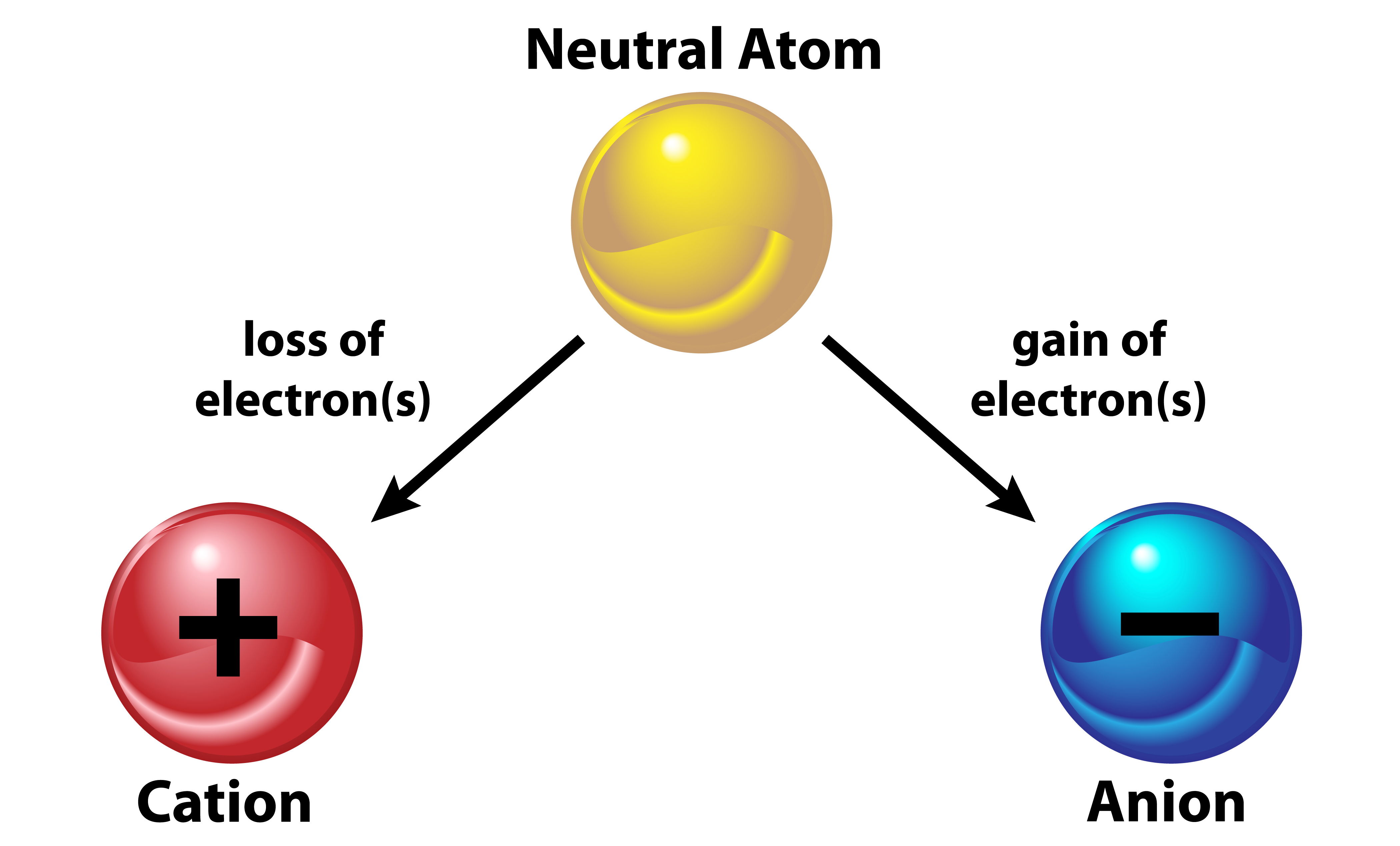
There are two main types of ions:
Cations: Cations are positively charged ions formed when atoms lose one or more electrons. The loss of electrons results in an excess of positive charge compared to the number of negative charges from protons, giving the ion a net positive charge. Cations are typically formed by metals and are named after the metal followed by the word "ion." For example:
- Sodium ion (Na⁺): Formed when a sodium atom loses one electron.
- Magnesium ion (Mg²⁺): Formed when a magnesium atom loses two electrons.
- Aluminum ion (Al³⁺): Formed when an aluminum atom loses three electrons.
Anions: Anions are negatively charged ions formed when atoms gain one or more electrons. The addition of electrons results in an excess of negative charge compared to the number of positive charges from protons, giving the ion a net negative charge. Anions are typically formed by non-metals and are named by replacing the ending of the element name with "-ide" followed by the word "ion." For example:
- Chloride ion (Cl⁻): Formed when a chlorine atom gains one electron.
- Oxide ion (O²⁻): Formed when an oxygen atom gains two electrons.
- Sulfide ion (S²⁻): Formed when a sulfur atom gains two electrons.
Ions play crucial roles in chemical reactions, solution chemistry, and various biological processes. They are involved in the formation of ionic compounds, electrolytes in solution, and the conduction of electricity in electrolytic cells. Understanding ions and their behavior is fundamental to understanding the behavior of matter at the atomic and molecular levels.
Polyatomic Ion A polyatomic ion is a charged species (ion) composed of two or more atoms covalently bonded together, with an overall positive or negative charge. Unlike simple ions formed from single atoms gaining or losing electrons, polyatomic ions consist of multiple atoms bonded together as a single unit and carry a net electrical charge.Polyatomic ions can be either positively charged (cations) or negatively charged (anions), depending on whether they lose or gain electrons, respectively. These ions often have distinct names and formulas, and they play essential roles in various chemical reactions and compounds.Some common examples of polyatomic ions include:
- Nitrate ion: NO₃⁻ (anion)
- Hydroxide ion: OH⁻ (anion)
- Ammonium ion: NH₄⁺ (cation)
- Carbonate ion: CO₃²⁻ (anion)
- Sulfate ion: SO₄²⁻ (anion)
- Phosphate ion: PO₄³⁻ (anion)
These polyatomic ions are found in numerous compounds and are involved in reactions such as acid-base reactions, precipitation reactions, and redox reactions. Understanding polyatomic ions is crucial in chemistry, as they contribute to the formation and properties of many compounds encountered in various chemical systems.